1.
Sources and Composition
1.1
Sources
Cordyceps is a mushroom traditionally used to treat sexual dysfunction and fertility in Chinese medicine, as well as a general sexual tonic and libido/performance enhancer.[1][2] This mushroom belongs to phylum Ascomycoa, the sub-phylum Ascomycotina and the class Clavicipataceae; which as a whole is seen as medicinal.[2]
There are multiple species of Cordyceps. The most commonly used species is Sinensis, which is a Cordyceps to be shown to contain the bioactive compound Cordycepin (3'-deoxyadenosine). It is also present in Militaris and Kyushuensis.[2][3] Isolation of Cordycepin dates back to 1950, first discovered in Militaris.[4] Cordycepin is known as a nucleotide analogue, due to its structural similarities to adenosine.[5]
Cordyceps possesses a potent anti-oxidative effect although its potency is quite variable from one sample to the next.[6] The anti-oxidative effects of Cordyceps come mostly from the polysaccharide content, and is equally potent between the Mycelium and the fruiting body of Cordyceps.[6]
1.2
Composition
Cordyceps, as a mushroom, contains a variety of compounds including:
- Cordycepin, seen as the main bioactive and also known as 3'-deoxyadenosine[7]
- Ergosterol and Ergosterol palmitate[8]
- 'Cordycepic Acid' which is just D-Mannitol,[9] but some related compounds such as 3,4-O-isopropylidene-d-mannitol[8]
- (Sinensis Mycelium) Glucosamine at 25.5mcg/mL (standard cultivation) and can be increased to 13.1-29.3mg/mL[10]
- (kyushuensis) Sodium (15,238mcg/g), Potassium (23,860mcg/g), Calcium (6248mcg/g), Magnesium (5630mcg/g), Iron (556mcg/g), and Zinc (32.9mcg/g)
- (In selenium-enriched grown mushrooms), selenomethionine, senelite, and selanate[11]
With some bioactive amines:
- Cordymin (amino acid sequence MAPPYGYRTPDAAQ)[12]
- 'CMP' from the fruiting bodies[13]
- Peptides containing alpha-aminoisobutyric acid[1] which have both water-soluble and water insoluble components with similar testosterone-boosting properties[14]
Bioactive polysaccharides
- F2, a water soluble polysaccharide at 13.46% dry weight or less[14]
- F3, poorly water soluble with a nitrogen content at 84.85% dry weight or less (this study did not use pure F3)[14]
- CS-PS (12kDa; mannose, rhamnose, arabinose, xylose, glucose and galactose at 38.37%, 2.51%, 2.21%, 5.22%, 27.44%, and 24.25% of total polysaccharide)[15]
- CME-1, A specific mannose:galactose 4:6 polysaccharide that is a spingomyelinase inhibitor[16]
- CMP polysaccharides (polysaccharide extract consisting of 65.4% dry weight of Cordyceps) which the main bioactive fragment, CMP-II (16.7% dry weight), having a 56.4:26.4:17.2 ratio of glucose:galatose:mannose and 89.48% of total weight being sugars[17]
- Another polysaccharide (CPS1) with glucose:galactose:mannose but in a ratio of 2.8:1:2.9 and 99% sugars by weight[18]
1.3
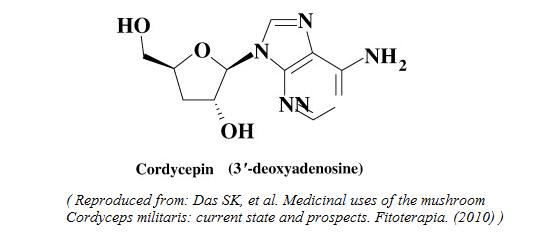
Structure of Cordycepin
Cordycepin is known as a nucleotide analogue due to its structural similarity to adenosine.
2.
Neurology
2.1
Fatigue
150-300mg/kg of the hot water extract of the Mycelia of Cordyceps Sinensis to rats orally (mostly carbohydrates), the time it took for rats to become fatigued during a swimming test increased to a similar degree with both doses and was approximately (value derived form graph) a 12.5% increase.[19]
2.2
Stress
Consumption of 150mg/kg of a hot water extract for a week in rats is associated with a lessening of biochemical markers of stress with reduction in total cholesterol (effectively normalized to non-stress control) and attenuations of the decline in the weight of the spleen (24%), adrenal (91%), and liver (37%).[19]
3.
Effects on Hormones
3.1
Testosterone
Cordyceps can reduce HcG and cAMP-stimulated steroidogenesis (via PKA and possibly inhibiting P450scc by 30%, the enzyme that converts cholesterol to pregnenolone).[20] This same study showed that Cordyceps did not reduce testosterone production when coincubated with androstenedione or pregnenolone, suggesting it does not influence enzymes in the later portion of steroidogenesis.[20] Interestingly, this study also showed that Cordyceps was able to inhibit Forskolin-induced steroidogenesis, which is cAMP-induced and how the herb Coleus Forskohlii increases testosterone.[20] This inhibition of testosterone synthesis stimulated by cAMP and HcG has been noted elsewhere,[21][22] and inhibition of PKA abolishes the effects of Cordyceps.[23]
In cell stimulated at the Luteinizing Hormone (LH) receptor, which normally induces steroidogenesis via a cAMP-dependent pathway, the incubation of Cordyceps can suppress this cascade
In cells not intentionally stimulated with HcG, Corcyceps extract reliably increases testosterone secretions from cells with an ideal concentration of 3mg/mL, shown in two separate studies on dose-response.[14][21] Concentrations greater than 10mg/mL are associated with declines in testosterone related to baseline.[21] Protein fragments in Cordyceps have been implicated in being the active compounds[14] although Cordycepin appears to be active as well.[23] Feeding isolated Cordycepin at 40mg/kg bodyweight does not increase testosterone in vivo, however, yet it does when injected suggesting poor bioavailability.[23]
Had the opposite effects in cells note treated via the LH receptor, and may increase testosterone secretion in these scenarios
The mycelium of Cordyceps Militaris at 1 or 5% of their diet by weight was shown to increase circulating testosterone levels in rats after 6 weeks of supplementation. [24] During this period, bodyweight did not significantly differ between groups, sperm content of the epididymus increased by 53% and 37% respectively to the 1% and 5% diets and motility increased by 31% and 19%; serum testosterone was increased to around 700pg/mL relative to control fluctuating just below 600pg/mL over the 6 week period. The peaks were erratic and demonstrated a trend to significant differences at 2 weeks in the 5% group, declining to baseline at 5 weeks, and then spiking up again to be significantly different at 6-8 weeks; 1% intake was relatively stable up to 5 weeks were it trended upwards and remained significantly different from control until cessation at 8 weeks.[24] Another study conducted in immature mice (without the influence of Luteinizing hormone, to stimulate central hypogonadism) found that 0.02-0.2g/kg bodyweight increased circulating testosterone relatively equally (3.83 and 3.69ng/ml, respectively) from a baseline level of 1.38+/-0.047ng/mL[14] while isolated water-soluble protein fragments required an intake of 0.2g/kg bodyweight.[14] Despite these studies being in immature rats, one study suggests that there are no differences in testosterone synthesis in immature and mature.[25]
More dramatic spikes are seen with intraperitoneal injections of cordycepin, which exhibits does-dependent increases in testosterone. 40mg/kg bodyweight over 7 days has been shown in mice to increase testosterone from 2.88+/-0.19pg/uL to 10.97+/-2.31pg/uL.[23] This spike was vicariously through an upregulation of A2a adenosine receptors (3-7 fold increase) with a concomitant decrease in A2b recpetor content.[23] Co-incubation with an A2a receptor antagonist (in this study, CSC was used; caffeine is a popular A2a antagonist) reduced the increase in testosterone by 20%, but inhibition of A3 reduced it by 50%.[23] The ultimate effect of Cordycepin is that it upregulates the StAR enzyme, which transporters cholesterol into the mitochondria for metabolism, a rate-limiting step of steroidogenesis.
Cordyceps appears to increase testosterone synthesis in rats, and has multiple compounds that could do this (protein fragments, Cordycepin); the protein fragments appear to be biologically relevant, as 40mg/kg Cordycepin ingested orally didn't do anything to testosterone in mice yet 0.2mg/kg whole Cordyceps did. Cordyceps may possess testosterone regulatory properties, rather than blind spiking of testosterone
3.2
Estrogen
Cordyceps Militaris supplementation was shown to increase estradiol levels in rats fed 1% or 5% of their diet as the mycelium, and although a significant spike was seen 2 weeks after supplementation (from 30pg/mL to the 60-70 range), it declined to baseline at 4 weeks and remained insignificantly different from control.[24]
3.3
Follicle-Stimulating Hormone
Despite increases in sperm content of rats, no significant influence on Follicle-Stimulating Hormone is seen with oral intake of 1 or 5% Cordyceps Militaris in the diet of male rats.[24]
3.4
Luteinizing Hormone
No significant effects are seen on circulating levels of luteinizing hormone in male rats after oral ingestion of 1% or 5% cordyceps militaris in the diet for 6 weeks.[24]
3.5
Prolactin
No significant effects on Prolactin are seen with either 1% or 5% dietary intake of Cordyceps Militaris over 6 weeks ingestion of the Mycelium.[24]
4.
Inflammation and Immunology
4.1
Macrophages
A polysaccharide from the fruiting bodies of Cordyceps Sinensis appears to have mitogenic properties on splenocytes as well as the ability to increase macrophage phagocytosis by 12% at 50-100mg/kg bodyweight,[15] although immunostimulatory polysaccharides are common to most species of Cordyceps. Immunostimulation has been found with Militaris (stimulation of macrophage Nitric Oxide and TNF-α)[17] and Militaris grown on Germinated soybeans.[26]
5.
Interactions with Oxidation
5.2
Interventions
After injection of D-Galactose into mice, which mimicks the effects of oxidation-mediated aging, Cordyceps Militaris supplementation was able to reduce oxidation via increasing the activity of anti-oxidative enzymes in the body; vicariously through the polysaccharide content.[27] These same effects have been seen with polysaccharides from Cordyceps Taii.[28] The anti-oxidant effects of Sinensis and Militaris are somewhat equivalent, with Sinensis slightly more potent.[29]
6.
Peripheral Organ Systems
6.1
Kidneys
Cordyceps has been traditionally used in chinese medicine for protective effects on renal tissue and for conditions such as chronic nephritis or pyelonephritis or general renal dysfunction.[30]
A concentration of 100mg/mL cordyceps (both c. sinensis and c. militaris) is able to suppress the proliferation of renal cells (mesangial cells) stimulated by LDL.[31]
Cordyceps has shown benefits in instances of kidney transplants inherently (injections of 0.5mL or 1.0mL of cordyceps an hour prior to the damaging stressor in the rat[32]) and synergistically with the immunosuppressant cyclosporin A at subtherapeutic doses of the latter[33] (a synergism that can extend to other organs[34]) thought to be tied into its own immunosuppressant and antiinflammatory properties, as there tends to be less immune cell infiltration of the tranplanted organ with the combination.[33][34] A lower maintenance dose of cyclosporin A has been noted in humans given cordyceps during the months after kidney replacement.[35]
There are two trials in humans undergoing renal transplants with cordyceps (c. sinensis)[36][35] where 1g of the supplement thrice daily alongside other immunosuppressants appeared to reduce urinary proteins and rates of chronic allograft neuropathies with cordyceps relative to control.[35] There was a reduced rate of organ toxicity seen during the following months with cordyceps (7.53%) relative to control (18.35%), although when assessing the liver enzyme ALT in patients without hepatoxocitiy there were no differences[35] and there are mixed results suggesting either a relative increase[36] or no differences[35] in surviviability compared to controls.
Cordyceps appears to be beneficial when given after kidney transplantation for reducing immune cell infiltration (a step which leads to damage and possible organ rejection) and when given to humans codyceps appears to have protective effects alongside standard immunosuppressive therapy
7.
Interactions with Cancer
7.1
Breast
In vitro, Cordycepin appears to induce apoptosis and reduce proliferation of breast cancer cells (MCF-7 and MDA-MB-231) with an approximate IC50 of 100uM.[37] Despite influencing both cell lines, the mechanisms appeared to differ.
In estrogen non-responsive cells (MDA-MB-231), Cordycepin appears to induce DNA fragmentation in a time and concentration dependent manner resulting in apoptosis. This appeared to be related to a release of cytochrome c from the mitochondria to the cytoplasm associated with caspase activation and PARP cleavage.[37] An aqueous extract of Coryceps per se shares these apoptotic effects associated with mitochondrial membrane depolarization, and aside from acting via Akt inhibition it is augmented with inhibition of PI3K/Akt in vitro.[38] Only one other study has noted anti-proliferative effects on this cell line, but was highly confounded with other Bioactive Mushrooms.[39]
In MCF-7 cells, the death of cells appeared to be autophagic.[37] Cordycepin failed to induce DNA fragmentation but 200uM clearly induced autophagic vacuoles and associated with conversion of LC3-I to LC3-II, commonly thought to be a biomarker for autophagy.[40] The exact mechanism was not elucidated but was independent of the estrogen receptors.[37] Beyond apoptotic, the ethanolic acetate fraction of Cordyceps (Mycelium) in general appears to have anti-proliferative effects on MCF-7 cells with an IC50 value of 44.7ug/mL (Petroleum 87.37+/-1.61ug/mL, ethanolic 79.57+/-2.68ug/mL, water ineffective).[9]
Another component, Cordymin (peptide) also appears to inhibit MCF-7 breast cancer proliferation in concentrations up to 5mg/mL but not surpassing 50% inhibition;[12] biological significance of this is unknown due to the large molecular weight (10,906Da) and being a long polypeptide possible not absorbed in vivo. Another peptide (12kDa) was able to induce cytotoxicity in MCF-7 cells and reduce their viability to 33.41+/-3.81% of control at 15uM with an IC50 of 9.3µM in vitro.[13]
Finally, in the highly invasive 4T1 cell line an injected water soluble extract of Cordyceps (10-50mg/kg) significantly inhibited metastasis as measured in the lung (when the tumors were injected into the breast of rodents) without significantly affecting tumor size whatsoever.[41] This study hypothesized that the immunostimulatory properties of Cordyceps on macrophages attenuated the rate of which 4T1 cells progressed from G0 /G1 to GM phase, which was demonstrated in vitro.[41]
A variety of compounds that could benefit breast cancer by reducing proliferation of cells or induce cancer cell death, but none of these mechanisms are currently established in living models or compared against active control drugs (to assess potency)
7.2
Leukemia
In comparing several fractions of Cordyceps Mycelium on HL-60 cells the ethanolic (87.57+/-1.69), ethanolic acetate (21.77+/-1.30) and petroleum (62.87+/-1.49) extracts but not water show some anti-proliferative effects with those respective IC50 values.[9]
7.3
Melanoma
In vitro, extracts of the Mycelium of Cordyceps appear to inhibit proliferation of Melanoma cells with IC50 values in B16 cells of 99.47+/-1.67ug/mL in ethanolic and ethanolic acetate 12.17+/-1.24ug/mL, with water and petroleum extracts being fairly ineffective.[9] Due to the potency of the ethyl acetate fraction, it was tested in mice implanted with B16 tumors at 0.05mg/kg (injections) and decreased tumor weight by 48% but underperformed relative to the active control of Cytoxan (62%).[9] When comparing bioactives of these extracts, the ethyl acetate appear to have a very large dose of ergosterol.[9]
7.4
Hepatocellular Carcinoma
In HepG2 cells, Cordyceps Mycelium shows some weak anti-proliferative effects with the ethanolic (84.27+/-1.32ug/mL), ethyl acetate (16.27+/-1.39ug/mL), and petroleum extracts (132.37+/-1.31ug/mL) and their respective IC50 values.[9]
7.5
Colorectal
A general Cordyceps extract appears to reduce proliferation of colon cancer cells (HT-29 and SW480) secondary to anti-inflammatory effects, preventing TNF-α induced NF-kB activity.[42]
When looking at specific bioactives of Cordyceps (this study used a Colon205 cell line), there were no remarkable IC50 values but some notable ones were Cordycepin (32.6+/-3.2ug/mL) and ergosterol palmitate (62.4+/-3.2ug/mL),[8] this study also suspected that the mechanisms were secondary to anti-inflammatory effects, these effects which have been noted in the colon previously in vivo.[43]
A bioactive peptide from Cordyceps (that has demonstrated activity against breast cancer cells) does not possess this same anti-proliferative efficacy against colon cancer cells[12] and both an n-butanol and chloroform extract of Cordyceps (Sinensis) failed to significantly reduce proliferation of Colon205 adenocarcinoma cells.[44] Neither of these studies were done in cultures with immune cell mediators.
May interact with the immune system (in a matter of suppression) to indirectly be anti-cancer, but actions in cell cultures are relatively lacklustre and no in vivo evidence exists currently
7.6
Bladder
One in vitro study using bladder cancer cells (5637 cell line) noted that 15uM of a peptide known as CMP was able to reduce viability to 39.06+/-15.60% of control with an IC50 of 8.1µM.[13] The mechanism of CMP was not established.
Cordycepin at an IC50 of 200uM was able to induce dose-dependent growth inhibition possibly via G2/M-phase arrest in both 5637 and T-24 cell lines alongside downregulation of various molecules associated with G2/M phase (pCdc25c and Cdc25c, pCdc2 and Cdc2, cyclin B1).[45] p27 and p53 did not appeared to be involved in this arrest, with JNK activation by Cordycepin appearing to mediate the beneficial effects.[45] A concurrent reduction in AP-1, NF-kB, and MMP-9 genomic activity may accompany Cordycepin's actions in bladder cancer cells.[46]
Possible anti-bladder cancer effects, but no in vivo evidence for efficacy or comparison to active control drugs
8.
Interactions with Longevity
8.1
Mechanisms
Cordyceps sinensis is thought to have antiaging properties due to improving antioxidant enzyme status in the brains of rats with accelerated senesence (induced by D-galactose).[47]
9.
Safety and Toxicology
9.1
General
References
- ^Ng TB, Wang HXPharmacological actions of Cordyceps, a prized folk medicineJ Pharm Pharmacol.(2005 Dec)
- ^Das SK, Masuda M, Sakurai A, Sakakibara MMedicinal uses of the mushroom Cordyceps militaris: current state and prospectsFitoterapia.(2010 Dec)
- ^Ling JY, Sun YJ, Zhang H, Lv P, Zhang CKMeasurement of cordycepin and adenosine in stroma of Cordyceps sp. by capillary zone electrophoresis (CZE)J Biosci Bioeng.(2002)
- ^CUNNINGHAM KG, MANSON W, SPRING FS, HUTCHINSON SACordycepin, a metabolic product isolated from cultures of Cordyceps militaris (Linn.) LinkNature.(1950 Dec 2)
- ^Ahn YJ, Park SJ, Lee SG, Shin SC, Choi DHCordycepin: selective growth inhibitor derived from liquid culture of Cordyceps militaris against Clostridium sppJ Agric Food Chem.(2000 Jul)
- ^Li SP, Li P, Dong TT, Tsim KWAnti-oxidation activity of different types of natural Cordyceps sinensis and cultured Cordyceps myceliaPhytomedicine.(2001 May)
- ^Li C, Yan A, Cai C, Liu ZFast determination of adenosine and cordycepin in Cordyceps and its deserted solid mediumSe Pu.(2012 Jul)
- ^Rao YK, Fang SH, Wu WS, Tzeng YMConstituents isolated from Cordyceps militaris suppress enhanced inflammatory mediator's production and human cancer cell proliferationJ Ethnopharmacol.(2010 Sep 15)
- ^Wu JY, Zhang QX, Leung PHInhibitory effects of ethyl acetate extract of Cordyceps sinensis mycelium on various cancer cells in culture and B16 melanoma in C57BL/6 micePhytomedicine.(2007 Jan)
- ^Choi JW, Ra KS, Kim SY, Yoon TJ, Yu KW, Shin KS, Lee SP, Suh HJEnhancement of anti-complementary and radical scavenging activities in the submerged culture of Cordyceps sinensis by addition of citrus peelBioresour Technol.(2010 Aug)
- ^Dong JZ, Ding J, Yu PZ, Lei C, Zheng XJ, Wang YComposition and distribution of the main active components in selenium-enriched fruit bodies of Cordyceps militaris linkFood Chem.(2013 Apr 15)
- ^Wong JH, Ng TB, Wang H, Sze SC, Zhang KY, Li Q, Lu XCordymin, an antifungal peptide from the medicinal fungus Cordyceps militarisPhytomedicine.(2011 Mar 15)
- ^Park BT, Na KH, Jung EC, Park JW, Kim HHAntifungal and Anticancer Activities of a Protein from the Mushroom Cordyceps militarisKorean J Physiol Pharmacol.(2009 Feb)
- ^Hsu CC, Huang YL, Tsai SJ, Sheu CC, Huang BMIn vivo and in vitro stimulatory effects of Cordyceps sinensis on testosterone production in mouse Leydig cellsLife Sci.(2003 Sep 5)
- ^Zhang J, Yu Y, Zhang Z, Ding Y, Dai X, Li YEffect of polysaccharide from cultured Cordyceps sinensis on immune function and anti-oxidation activity of mice exposed to 60CoInt Immunopharmacol.(2011 Dec)
- ^Wang SH, Yang WB, Liu YC, Chiu YH, Chen CT, Kao PF, Lin CMA potent sphingomyelinase inhibitor from Cordyceps mycelia contributes its cytoprotective effect against oxidative stress in macrophagesJ Lipid Res.(2011 Mar)
- ^Lee JS, Kwon JS, Won DP, Lee JH, Lee KE, Lee SY, Hong EKStudy of macrophage activation and structural characteristics of purified polysaccharide from the fruiting body of Cordyceps militarisJ Microbiol Biotechnol.(2010 Jul)
- ^Wang Y, Wang M, Ling Y, Fan W, Wang Y, Yin HStructural determination and antioxidant activity of a polysaccharide from the fruiting bodies of cultured Cordyceps sinensisAm J Chin Med.(2009)
- ^Koh JH, Kim KM, Kim JM, Song JC, Suh HJAntifatigue and antistress effect of the hot-water fraction from mycelia of Cordyceps sinensisBiol Pharm Bull.(2003 May)
- ^Hsu CC, Tsai SJ, Huang YL, Huang BMRegulatory mechanism of Cordyceps sinensis mycelium on mouse Leydig cell steroidogenesisFEBS Lett.(2003 May 22)
- ^Huang BM, Hsu CC, Tsai SJ, Sheu CC, Leu SFEffects of Cordyceps sinensis on testosterone production in normal mouse Leydig cellsLife Sci.(2001 Oct 19)
- ^Wong KL, So EC, Chen CC, Wu RS, Huang BMRegulation of steroidogenesis by Cordyceps sinensis mycelium extracted fractions with (hCG) treatment in mouse Leydig cellsArch Androl.(2007 Mar-Apr)
- ^Leu SF, Poon SL, Pao HY, Huang BMThe in vivo and in vitro stimulatory effects of cordycepin on mouse leydig cell steroidogenesisBiosci Biotechnol Biochem.(2011)
- ^Chang Y, Jeng KC, Huang KF, Lee YC, Hou CW, Chen KH, Cheng FY, Liao JW, Chen YSEffect of Cordyceps militaris supplementation on sperm production, sperm motility and hormones in Sprague-Dawley ratsAm J Chin Med.(2008)
- ^Huang YL, Leu SF, Liu BC, Sheu CC, Huang BMIn vivo stimulatory effect of Cordyceps sinensis mycelium and its fractions on reproductive functions in male mouseLife Sci.(2004 Jul 16)
- ^Ohta Y, Lee JB, Hayashi K, Fujita A, Park DK, Hayashi TIn vivo anti-influenza virus activity of an immunomodulatory acidic polysaccharide isolated from Cordyceps militaris grown on germinated soybeansJ Agric Food Chem.(2007 Dec 12)
- ^Li XT, Li HC, Li CB, Dou DQ, Gao MBProtective effects on mitochondria and anti-aging activity of polysaccharides from cultivated fruiting bodies of Cordyceps militarisAm J Chin Med.(2010)
- ^Xiao JH, Xiao DM, Chen DX, Xiao Y, Liang ZQ, Zhong JJPolysaccharides from the Medicinal Mushroom Cordyceps taii Show Antioxidant and Immunoenhancing Activities in a D-Galactose-Induced Aging Mouse ModelEvid Based Complement Alternat Med.(2012)
- ^Yu HM, Wang BS, Huang SC, Duh PDComparison of protective effects between cultured Cordyceps militaris and natural Cordyceps sinensis against oxidative damageJ Agric Food Chem.(2006 Apr 19)
- ^Cordyceps as an Herbal Drug
- ^Zhao-Long W1, Xiao-Xia W, Wei-Ying CInhibitory effect of Cordyceps sinensis and Cordyceps militaris on human glomerular mesangial cell proliferation induced by native LDLCell Biochem Funct.(2000 Jun)
- ^Shahed AR1, Kim SI, Shoskes DADown-regulation of apoptotic and inflammatory genes by Cordyceps sinensis extract in rat kidney following ischemia/reperfusionTransplant Proc.(2001 Sep)
- ^Ding C1, Tian P, Jia L, Li Y, Ding X, Xiang H, Xue W, Zhao YThe synergistic effects of C. Sinensis with CsA in preventing allograft rejectionFront Biosci (Landmark Ed).(2009 Jan 1)
- ^Jordan JL1, Hirsch GM, Lee TDC. sinensis ablates allograft vasculopathy when used as an adjuvant therapy with cyclosporin ATranspl Immunol.(2008 Jul)
- ^Li Y1, Xue WJ, Tian PX, Ding XM, Yan H, Pan XM, Feng XSClinical application of Cordyceps sinensis on immunosuppressive therapy in renal transplantationTransplant Proc.(2009 Jun)
- ^Sun M1, Yang YR, Lu YP, Gao R, Wang L, Wang J, Tang KClinical study on application of bailing capsule after renal transplantationZhongguo Zhong Xi Yi Jie He Za Zhi.(2004 Sep)
- ^Choi S, Lim MH, Kim KM, Jeon BH, Song WO, Kim TWCordycepin-induced apoptosis and autophagy in breast cancer cells are independent of the estrogen receptorToxicol Appl Pharmacol.(2011 Dec 1)
- ^Jin CY, Kim GY, Choi YHInduction of apoptosis by aqueous extract of Cordyceps militaris through activation of caspases and inactivation of Akt in human breast cancer MDA-MB-231 CellsJ Microbiol Biotechnol.(2008 Dec)
- ^Jiang J, Sliva DNovel medicinal mushroom blend suppresses growth and invasiveness of human breast cancer cellsInt J Oncol.(2010 Dec)
- ^Bommareddy A, Hahm ER, Xiao D, Powolny AA, Fisher AL, Jiang Y, Singh SVAtg5 regulates phenethyl isothiocyanate-induced autophagic and apoptotic cell death in human prostate cancer cellsCancer Res.(2009 Apr 15)
- ^Jordan JL, Nowak A, Lee TDActivation of innate immunity to reduce lung metastases in breast cancerCancer Immunol Immunother.(2010 May)
- ^Huang H, Wang H, Luo RCInhibitory effects of cordyceps extract on growth of colon cancer cellsZhong Yao Cai.(2007 Mar)
- ^Han ES, Oh JY, Park HJCordyceps militaris extract suppresses dextran sodium sulfate-induced acute colitis in mice and production of inflammatory mediators from macrophages and mast cellsJ Ethnopharmacol.(2011 Apr 12)
- ^Rao YK, Fang SH, Tzeng YMEvaluation of the anti-inflammatory and anti-proliferation tumoral cells activities of Antrodia camphorata, Cordyceps sinensis, and Cinnamomum osmophloeum bark extractsJ Ethnopharmacol.(2007 Oct 8)
- ^Lee SJ, Kim SK, Choi WS, Kim WJ, Moon SKCordycepin causes p21WAF1-mediated G2/M cell-cycle arrest by regulating c-Jun N-terminal kinase activation in human bladder cancer cellsArch Biochem Biophys.(2009 Oct 15)
- ^Lee EJ, Kim WJ, Moon SKCordycepin suppresses TNF-alpha-induced invasion, migration and matrix metalloproteinase-9 expression in human bladder cancer cellsPhytother Res.(2010 Dec)
- ^Ji DB, Ye J, Li CL, Wang YH, Zhao J, Cai SQAntiaging effect of Cordyceps sinensis extractPhytother Res.(2009)




