Sensing caloric density: can we eat less if we eat more?
Can preloading with low energy density foods eaten slightly before a meal reduce overall food intake?
Last Updated: August 18, 2020
Become an Examine+ member to get full access to the Examine Database, Study Summaries, and Supplement Guides.
Try for free for 7 days and stay on top of the latest research.
It is often surprising—and hugely unfair—how few calories are needed for weight gain. Even a seemingly negligible change in caloric intake of 3-4% can result in a one kilogram increase in weight per year[1] in adults. Exploring the factors that contribute to short-term caloric intake allows for improved understanding into the drivers of appetite, practical strategies for reducing energy consumption, and obesity prevention.
One way to contribute to caloric reduction in the long run is by reducing caloric intake in the short term. Since it can be challenging to make an accurate assessment[2] of caloric intake, examining eating episodes in a laboratory setting is an important strategy for gaining a greater understanding of the subtle factors that can contribute to caloric intake. The types of foods eaten before the main meal can broadly be described as a ‘preload’. A preload is a fixed amount of a predefined food, eaten before consumption of an actual meal[3]. The impact the preload has on a subsequent meal (or snack) is measured, therefore allowing greater insight into the drivers of short-term caloric intake. One thing which could impact the influence of a preload on the following meal is its energy density.
The concept of energy density is illustrated in Figure 1. It’s defined as the energy (calories) each gram of food delivers. Energy density can be an indication of food quality[4]. Fat, which delivers nine kcal per gram[5], has the most potent impact on energy density compared to carbohydrate or protein (which each deliver about four kcal per gram). Foods high in fat are considered to have high energy density (HED). Contrary to the idea that energy density encourages satiety, the opposite may be true: given that HED foods often taste pleasant, it has been suggested that palatability disrupts satiety signaling[6], therefore promoting overeating.
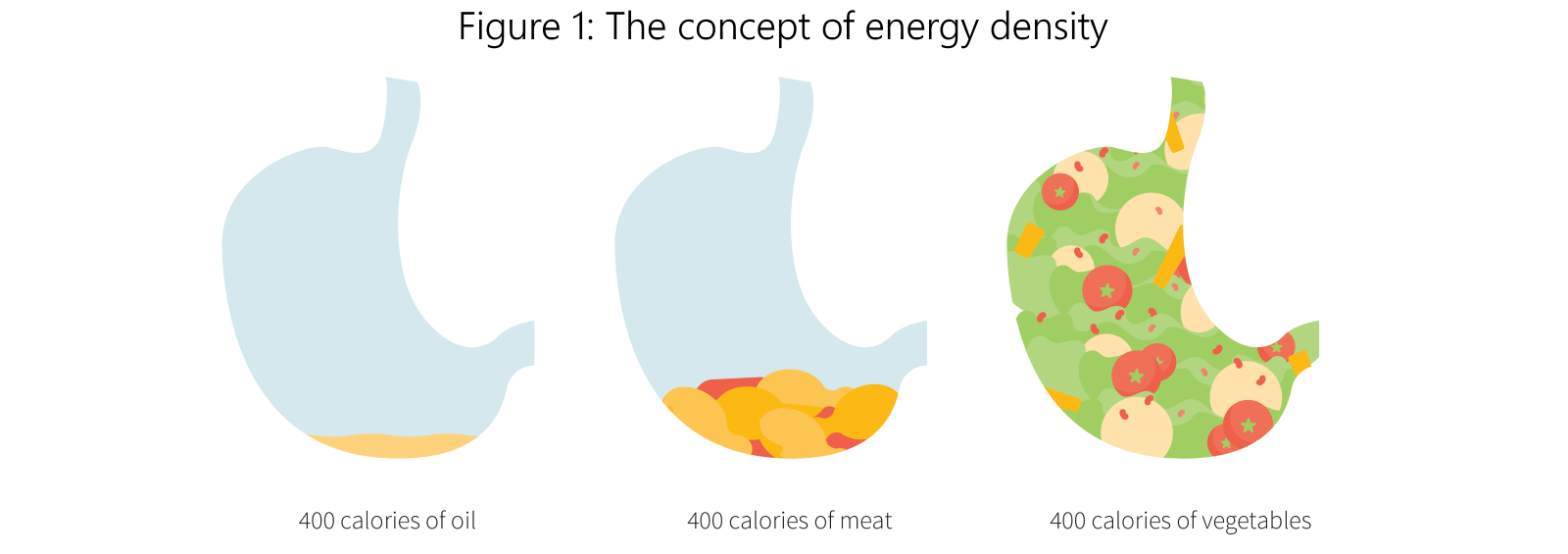
The energy density of any food—whether a preload or meal—is an important factor to consider, as diets high in energy density have been shown to contribute to both weight gain and obesity[7]. Yet, whether energy density is linked with satiety[8] is not entirely clear. There is not a linear relationship between calories consumed and degree of satiety experienced, and for this reason satiety signaling’s relationship with the energy density of foods has become an urgent area of focus within obesity research.
Typically, foods that are low energy density (LED) contain higher amounts of water. This is the case with fruits and vegetables. Dietitians and medical professionals often recommend incorporating a higher ratio of LED foods, such as fruits[9], vegetables[10], and soups[11], not only as a way to improve diet quality, but also a means to reduce caloric intake. This is because the higher water content in these foods contributes to both increased satiety and slower gastric emptying, thereby leading towards an overall reduction in [12]caloric intake[12].
In order to gain the clearest understanding of the ways these kinds of foods promote satiety, and therefore control of caloric intake, scientists have investigated the consequences of HED vs. LED preloads. The authors of the study under review sought to methodically review and meta-analyze the literature to date to determine the effect that preloads, and their HED vs. LED energy density, have on subsequent food intake.
The use of low energy density (LED) foods and preloads have been suggested as a means to reduce overall energy intake. However, studies to date show conflicting results in regard to whether LED preloads reduce subsequent energy intake and the role that preload energy density plays in satiety. The study under review is a meta-analysis of clinical trials assessing the effect of preload energy density on energy intake at a subsequent meal.
This was a meta-analysis of clinical trials involving people aged 2-60 years and without eating disorders. Two types of studies were investigated: those that looked at how modifying a preload shortly before a meal affected energy intake at that meal, and those that looked at how modifying one or more meals during the day affected energy intake at subsequent meals or snacks.
Ultimately, 35 crossover studies involving 1,273 participants were included in this meta-analysis. Nine studies were conducted with children (3.5-9 years) and the remainder in adults (21-46.5 years). About half (n=18) the studies manipulated energy density by modifying either water or fat content, nine studies modified carbohydrate content, one study modified fiber content, and two studies increased fruit and vegetable intake. Six studies used other methods or did not indicate how energy density was manipulated.
This was a meta-analysis of 35 crossover trials and 1,273 participants investigating the effect of preload and meal energy density on subsequent food intake. A variety of preload paradigms were used, including manipulations of carbohydrate, fiber, fat, and water content. Studies involved both adults (75%) and children (25%).
This meta-analysis tells us that an LED preload consumed shortly before a meal significantly reduces energy intake at the meal compared to a preload with higher energy density. However, energy intake appeared to depend on the type of preload manipulation, with reductions observed in trials increasing fiber and water content, increases in trials reducing carbohydrate content, and no change in trials manipulating fat content. This is in line with mechanistic considerations, since foods high in fiber and water content lead to slower gastric emptying, intestinal transit time, and also the absorption of fat and carbohydrate[13]. So, this meta-analysis suggests that certain preloads, such as a non-cream soup right before a meal, are a good way to stave off unwanted additional calories from a main entree.
It should be emphasized, though, that timing might be key. The finding that LED foods curb appetite seemed to be contingent upon a temporal delay, since preloads reduced energy intake at subsequent meals but manipulating the energy density of meals did not have a notable effect on energy intake at later meals.
Interestingly, the authors found that LED preloads with lower carbohydrate content increased energy intake compared to high carbohydrate, HED preloads. This may be due to the postprandial consequences of simple carbohydrate consumption. The authors duly note that researchers rely on maltodextrin[14] to change the macronutrient makeup (maltodextrin is a simple carbohydrate). Since several satiety-based hormones respond immediately to an increase in carbohydrate (such as insulin and CCK), it may be the case that the HED preloads temporarily evoked satiety due to the hormonal responses to carbohydrate, not necessarily the satiating effects of the preload.
One key strength of this study was that the authors controlled for many of the features of the preloads and meals to control for possible confounding factors. For example, they looked at matching energy density, volume, and macronutrient composition of the foods used for preloads. Compared to a previous similar investigation[15], the authors of the current study placed more emphasis on analysis of energy content of the preloaded foods, particularly with regard to the macronutrient content. For this reason, it appears to be the most detailed meta-analysis of preload data to date.
Nonetheless, there are limitations. Energy, macronutrient content, and texture of the foods were all taken into account, but palatability[6] was not. Palatability has been shown to have tremendous influence[16] on short and long term caloric intake. Also, there was no investigation of food temperature, which has also been shown to affect short term caloric intake[17]. Most notably, there was no specific definition of ‘high energy density’ versus ‘low energy density’ for the foods, as different studies relied on different cut-off points. This latter point makes generalization tremendously difficult. The LED preloads and meals contained 0.07-3.12 kcal/g, compared to 0.49-4.4 kcal/g in HED preloads and meals.
Finally, this meta-analysis does not provide data on long-term effects of using preloads. Palatability, temperature, and drives toward homeostasis may outweigh the short term effect of a preload given before a single meal. Accordingly, it is not appropriate to make conclusions about long-term effects on energy intake or body weight based on this study’s finding that preloads reduce subsequent meal energy intake.
Ultimately, it was found that preloads served directly before a meal that were high in fiber and water content (but low in energy) had the most significant effect on appetite reduction. Shorter temporal delays (i.e., those conditions where the preload was served directly before the meal) also had a significant impact on reducing short term intake. The heterogeneity between the studies, and the lack of consistency in determining low vs. high energy limit the ability to generalize the results of this study, though.
Satiety signaling plays a central role in eating motivation, so understanding satiety can lead to greater understanding of obesity prevention. Unfortunately, people cannot trust satiety signaling alone to guide their food intake. As shown in Figure 2, highly palatable food can exploit feeding motivation and contribute directly to weight gain and obesity. Investigating satiety and the subtle differences between energy density, temporal delay, and the macronutrient content of food is essential within both obesity and ingestive behavior research. Preload paradigms provide a platform for understanding how HED or LED foods contribute towards, or inhibit, satiety.

Satiation leads to the termination of eating, and satiety is the result of prior food ingestion[18]. While physiological processes have a significant influence on energy balance, it has become increasingly clear that eating behavior is also strongly influenced by non-homeostatic signals, such as food availability and palatability[19]. Moreover, environmental cues associated with rewarding foods can override homeostatic controls.
While it has been demonstrated that people are generally consistent in the weight of food[3] they consume, the precise mechanism that controls satiety is not fully understood. Given the complexity of the interplay between gut and cerebral hormones, the role of pleasure affiliated with food, and the difficulty associated with retraining food habits, it may be a while until researchers have a full grasp of the way foods (especially those that are HED, and immediately pleasurable) affect appetitive drive.
While the details aren’t fully sussed out, it’s pretty certain that the gut plays a big role in appetite, since many of the neural and hormonal signals of energy balance, and therefore satiety and hunger, start there. The gut also controls secretion of insulin and digestive enzymes[20]. Stimulating the gut with a high volume of LED foods, as opposed to an isocaloric low volume of HED foods, may provide sufficient lag time to allow the brain to respond by terminating the desire for food.
Other research has found that while some lag time between preload and the next feeding is important, you can have too much. In an independent meta-regression[21] of properties affiliated with a reduction of energy intake when ingesting a preload, researchers found the two strongest predictors of subsequent caloric intake were the physical state of the preload (specifically, solid and semi-solid foods), and the duration of intermeal interval (IMI) time. The reduction in energy intake was maximised when the IMI was between 30-120 minutes.
It’s not just timing between feedings that can contribute to satiety. The time it takes to consume the meal also plays a role. This is known as ‘slow spaced eating[22]’, or, in less fancy terms, chewing and eating food more slowly[22]. In healthy participants, researchers have shown slower chewing/slow spaced eating may serve to increase[23] postprandial response of appetite-inhibiting peptides PYY and GLP-1 in healthy participants. More recent data has shown slow spaced eating[22] also improves hunger and satiety ratings in obese participants with type 2 diabetes, providing a second strategy for enhancing satiety to reduce caloric intake.
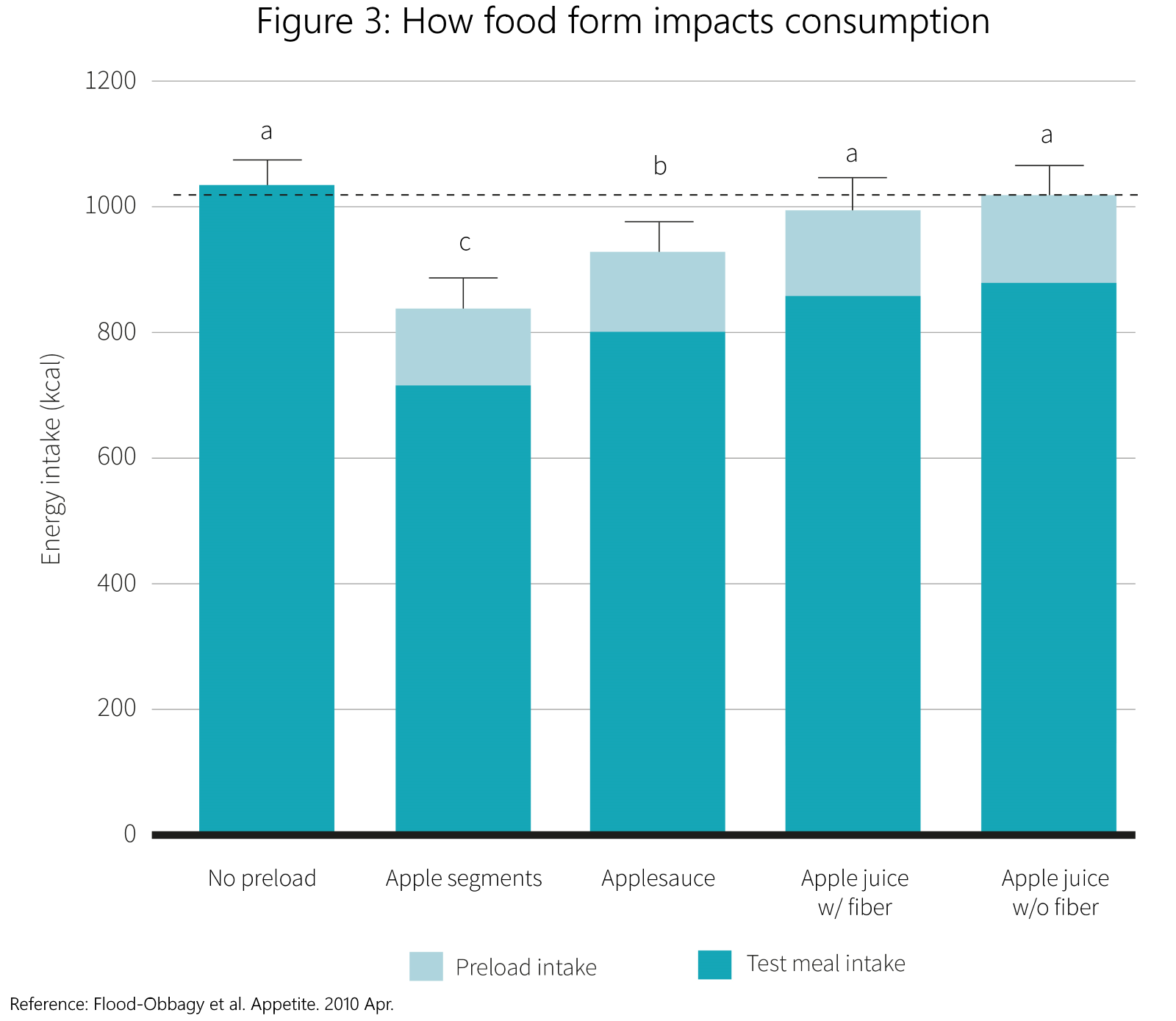
The findings of the meta-analysis under review corroborate other research in this area. For example, researchers have found significant reduction in energy intake when preload and test meals are infused with water, and researchers have found significant reduction in energy intake when participants consume whole foods[9] such as apples (versus energy- and fiber-matched sauces or juices), or even using salad as a preload[15] before an ad libitum meal. The data on how the form of food impacts consumption is shown in Figure 3. Finally, researchers have found that energy-matched soups lead to a significant reduction in caloric intake[11], compared to energy-matched foods, corroborating the idea that lower energy density and higher water content can lead to a reduction in overall energy intake.
The mechanisms of satiety signaling are not fully understood, but it involves a complex interplay involving physiological as well as psychological and environmental factors. Other research has demonstrated that eating food more slowly, and eating an LED preload containing solid or whole foods with a high water content 30 to 120 minutes before the next meal may be useful ways of reducing caloric intake.
Q. How are satiety signaling and obesity related?
Recent brain research has shown that, compared to lean controls, people with obesity have blunted responses toward satiety[24]. For example, both obese men and women have been shown to have weaker responses to meal termination cues (and satiety)[25], which has been attributed to decreased activation in the dorsolateral prefrontal cortex, an area in the brain associated with inhibitory response. Obese participants also show altered responses to gut-brain hormones which are linked with increase in appetite, such as ghrelin[26]. For example, ghrelin — which spikes during the anticipation for a meal, but is reduced quickly after eating — has been shown to be higher in obese participants[26] compared to lean controls.
In addition to reduced sensitivity to satiety signalling[27], it has been shown that individuals with obesity also demonstrate pronounced motivation for highly palatable food[28]. Researchers have suggested the increased motivation (or, ‘wanting’) for highly palatable foods sets off a motivational cascade that works like a vicious cycle, as physiological hunger signals (such as satiety) are compromised in those who struggle to maintain a lean body weight. Together, these features contribute to the biopsychological loop that that sustains both motivation for unhealthy foods, in addition to the ability to overeat more of them.
An extreme version of the way increased reward sensitivity and satiety insensitivity contribute towards weight gain can be viewed in those who suffer from a condition known as Prader-Willi Syndrome (PWS). PWS is a genetic disorder, characterized by extreme overeating (hyperphagia), intellectual impairment, and obesity. Left untreated, PWS will contribute to morbid obesity, and has served as a model for studying the brain and obesity. In brain imaging studies, researchers have found individuals who suffer PWS show increased activation[29] in regions in the brain associated with food wanting (the limbic region, including nucleus accumbens and amygdala), and decreased activation[29] in areas affiliated with inhibitory responses (such as the orbital and prefrontal cortices). The increased activation from the limbic region paired with muted activation of the frontal regions has been described as ‘bottom-up processing’. The opposite would be ‘top-down’ processing whereby the individual exercises appropriate inhibitory control of eating motivation and decisions. In addition to the tendency towards bottom-up processing, individuals with PWS have been shown to have higher fasting ghrelin[30]. The combination between increased gut hormones that would extend eating episodes, paired with the tendency towards bottom-up processing, describes the foundation for overeating behavior.
Q. Have researchers ranked foods according to how satiating they are?
Yes. Significant research has been devoted to underpinning a foods capacity for satiety. The satiety index[31] was originally created to compare a list of common foods to white bread (bread served as the reference point), with researchers categorizing foods based not only on their macronutrient components, but also hedonic and nutritional properties as well. In the original investigation, researchers created six categories (fruits, bakery products, snack foods, carbohydrate-rich foods, protein-rich foods, breakfast cereals) However, more recently, mapping satiety has become a focus within obesity and eating behavior research, and this original model has been expanded[32]. In line with the study under review, more satiating foods tend to have low caloric density. Also, lower fat percentage, higher protein percentage, and fiber content is correlated with a food being more satiating. In addition to food’s physical properties, this more recent satiety model also found that some psychological variables play a role in perceived satiation, including cost of the food and how healthy the food is perceived to be.
The meta-analysis under review provided further support for the role low energy density (LED) foods can have in reducing short term food intake. When eaten right before a meal, LED foods can reduce energy intake by about 100 kcal, provided they are both high in fiber and water content. The main effect of an LED preload can be attributed to a higher degree of satiety, and physiological effects such as slowing gastric emptying. Thus, as counter-intuitive as it might sound, eating more food before a meal—especially soup, salad, or a whole piece of fruit—may be an important strategy for reducing overall intake.