Deeper Dive: Shining a light on the effects of vitamin D on fall risk in older adults
This study suggests that higher vitamin D dosing doesn't prevent falls compared to lower dosing. Instead, it may cause more.
Last Updated: June 21, 2023
Become an Examine+ member to get full access to the Examine Database, Study Summaries, and Supplement Guides.
Try for free for 7 days and stay on top of the latest research.
An estimated one in four (28.7%) U.S. adults older than 65 had a fall in 2014[1]. One in four (25%) of those falls resulted in an injury. Falls among older adults ultimately resulted in nearly $32 billion in related medical expenses in 2015[2]. Alleviating this burden represents an opportunity to foster the health and safety of older people and decrease medical expenses. This is, however, a complex problem.
Vitamin D has been suggested as a primary target for intervention for two main reasons. First, it plays a critical role in bone formation, healthy bone structure, and bone mineral density. These effects on bone integrity could mitigate the damage done by falls when they occur. (Read more about fractures and vitamin D in this March 2018 Study Deep Dives article.) Vitamin D is also essential for muscle function, suggesting it could play a role in preventing falls in the first place by maintaining strength, balance, and coordination[3]. However results from a 2018 clinical trial in post-menopausal women suggests that higher dose (2,800 IU) supplementation might actually decrease muscle strength[4] (read the Mini here).
Second, vitamin D deficiency and insufficiency is widespread, with 28.9% of U.S. adults meeting the Endocrine Society’s criteria for deficiency (25(OH)D of less than 50 nmol/L) and 41.4% of adults considered to have insufficient levels[5] (25(OH)D levels (50–75 nmol/L). Furthermore, adults older than 60 years old are around 63% more likely to have vitamin D deficiency and 46% more likely to have vitamin D insufficiency than young adults[5]. This age-related decrease in vitamin D status may be the result of decreased synthesis from ultraviolet light[6], decreased mobility and thus decreased exposure to ultraviolet light[7], decreased vitamin D receptor expression[8], and inadequate dietary vitamin D intake[9]. Overall, the high prevalence of vitamin D insufficiency and deficiency in high-risk older populations means that plenty of people could potentially benefit from supplementation.
It’s reasonable to say, based on previous research, that vitamin D supplementation in the context of severe vitamin D deficiency (less than 20–25 nmol/L) is important for modifying the risk of falls[10], muscle function[11], and improving bone mineral density[12]. But what about the efficacy of supplementing in a population with marginal deficiency or insufficiency, i.e. people who probably don’t need high doses of vitamin D to achieve adequacy? Unfortunately, the results of numerous individual studies are mixed, with some studies suggesting mild benefit, others no benefit, and still others implying potential harm.
For example, one nine-month RCT testing the effects of 150,000 IU of vitamin D3 supplementation every three months in older, community-dwelling, postmenopausal women showed no differences between vitamin D and placebo on adverse effects, falls, mobility, or muscle strength[13]. A three-year RCT testing 800 IU of D3 daily with 500 mg of calcium vs. placebo in community dwelling older men and women showed that supplementation reduced fall risk in women, especially those with low baseline physical activity, but did not show a significant effect on men[14]. Another study of note showed that among older community-dwelling women, annual oral administration of 500,000 IU of D3 resulted in an increased risk of falls and fractures[15], confirming the lackluster or sometimes detrimental effects of vitamin D supplements on fall risk.
One comparative review has attempted to clarify why different meta-analyses of vitamin D and fall risk come to different conclusions. The authors conclude that it comes down to issues that could be found in any meta-analysis: differences in study inclusion/exclusion, differences in data extraction, and differences in statistical analysis[16].
When it comes to falls and frailty, all the evidence points to a U-shaped relationship between vitamin D levels and fall risk, similar to what researchers have noted for cancer, cardiovascular disease[17], and fractures[18]. Supplementing with vitamin D to adequacy appears to significantly improve fall risk, but supplementing beyond adequacy may do more harm than good.
Prior to the initiation of the present study in 2015, there had been a number of recommendations for vitamin D supplementation for fall prevention in older adults, each based on extensive reviews. Even though each set of guidelines aims to summarize the totality of clinical evidence, each one arrived at a different, sometimes substantially different, conclusion. The main takeaways of these guidelines are laid out in Figure 1.
| Organization | Blood level targets | Dose of supplemental vitamin D | Applicable population |
|---|---|---|---|
| American Geriatrics Society Workgroup on Vitamin D Supplementation for Older Adults[19] | 25(OH)D concentration of 75 nmol/L (30 ng/mL) as a minimum goal. | At least 1,000 IU daily alongside calcium. | Everyone over 65 years old, whether or not they live independently or in an institutionalized setting. |
| Endocrine Society[20] | States that an exact target is somewhat controversial, but defers to the standard definitions of 25(OH)D) levels of 50 nmol/L (20 ng/mL) indicating deficiency, and 75 nmol/L (30 ng/mL) indicating insufficiency. | At least 600–800 IU daily, but up to 2,000 IU daily may be needed to raise 20(OH)D to sufficient levels. | 600 IU is the lowest dose for people ages 50–70, an 800 IU for people 70 and above. |
| International Osteoporosis Foundation[21] | 25(OH)D concentration of 75 nmol/L (30 ng/mL) as a minimum goal. | 800–1,000 IU daily would make many, but not all, people vitamin D sufficient. But since higher doses haven’t been established to prevent falls, they don’t necessarily recommend higher doses for all older adults. | People ages 60–65 years and up. Doses can be adjusted up or down based on sun exposure (down) or obesity status (up). |
| US Preventive Services Task Force[10] | Only to prevent deficiency (25(OH)D concentration below 50 nmol/L or 20 ng/mL). | Not applicable: This group recommends against the routine use of vitamin D supplementation to prevent falls or fractures. | Community-dwelling people over 65 years without osteoporosis and without vitamin D deficiency. |
| Institute of Medicine[22] | Not applicable for fall prevention. | Insufficient evidence for fall prevention. This group recommends sticking with the RDA of 800 IU daily intake for this age group, supplementing as needed to achieve that intake level. | Adults 70 years and older. |
As a result of the suggestive, but ultimately inconclusive, research around vitamin D supplementation and fall risk, the National Institute on Aging (NIA) funded a clinical trial to assess the dose-response relationship between vitamin D ranging from the minimum dose necessary for adequacy to the upper limit of official recommendations and falls in a high-risk population.
Vitamin D deficiency is associated with poor bone health, poor muscle health, general frailty, and increased risk for falls, yet it is not entirely clear whether this relationship is causal. Vitamin D supplementation has been recommended for preventing falls in older adults by a number of organizations, and recommended against by others, reflecting the inconsistent results seen in clinical trials. This study, funded by the National Institute of Aging, aimed to clarify the effect of different doses of vitamin D on risk of falls in community-dwelling older adults with baseline deficient/insufficient vitamin D status.
The primary outcome of the preregistered “Study To Understand Fall Reduction and Vitamin D in You” (STURDY) trial was time to first fall or death over two years. Participants were included if they were community-dwelling residents of Hagerstown or Woodlawn, Maryland, 70 years of age or older, had elevated fall risk based on self-reported previous falls or presence of other risk factors for falls, and had a serum 25-hydroxyvitamin D (25(OH)D) level of 25 to 72.5 nmol/L. For reference, Woodlawn and Hagerstown are both at about 39° latitude, with about 105 clear, sunny days per year. Participants were not eligible if they had a cognitive impairment, abnormal calcium levels, kidney stones, or were already supplementing with daily vitamin D doses higher than 1,000 IU or calcium doses higher than 1,200 mg.
By the end of the study, 677 of the 688 enrolled participants were considered to have completed the trial. Average age at enrollment was 77.2 years, 56.4% of the participants were men, 18.2% identified as Black, and the mean 25(OH)D level was 55.3 nmol/L. In addition, 71.2% of participants were frail or had pre-frail status, and more than half had substantial functional impairment, which explains why 65.4% had fallen one or more times in the prior year. People with severe vitamin D deficiency (less than 25 nmol/L) were excluded.
The basics of the study design are laid out in Figure 2. This adaptive trial was divided between a dose-finding stage to determine the optimal dose of vitamin D, and a second stage to evaluate the primary outcome. In the first stage of the trial, participants were randomly assigned to 200 (control), 1,000, 2,000, or 4,000 IU of vitamin D3 per day. The first phase was set to conclude after the target sample size of 1,200 people were enrolled or once the probability that a specific non-control dose was superior to the others rose to 95%.
After two years and four months from the start of the trial, the best dose was identified as 1,000 IU per day, according to prespecified interim analysis, and participants were subsequently randomly assigned to the best dose or the control dose. The trial continued until the next interim analysis determined that there would be no point in continuing the study.
Figure 2: The basics of the adaptive study design

This study was designed to test the effects of different doses of vitamin D on the rate of falling or death in high-risk people over 70. The researchers accomplished this by using an adaptive randomized controlled trial design. This design has two parts. The first part identified which dose of vitamin D (1,000, 2,000, or 4,000 IU per day) was associated with the fewest falls or other adverse events compared to the control arm of 200 IU per day (a dose that helps most people achieve sufficient levels). Once the best dose was identified, participants in non-control arms were reassigned to the best dose, and new participants were randomized to the best dose or the control dose.
Participants were largely adherent to the intervention and total 25(OH)D levels increased progressively across the dosing groups initially and persisted through the second half of the study.
The initial dose-finding stage of the trial was halted early for two reasons. First, the 1,000 IU dose had been consistently predicted to be the best dose. Second, at this time point the rate of hospitalization or death was 2.69 times higher in the 2,000 IU per day group than the rate among those in the 200 IU per day group (33.6 per 100 vs. 12.1 per 100 person-years; hazard ratio: 2.69 with a 95% CI: 1.50 to 4.82). The 4,000 IU per day group also had a higher rate of hospitalization or death, although not a statistically significant one (hazard ratio: 1.68 with a 95% CI: 0.86 to 3.27). At the same time, compared to the low-dose control group, the primary composite outcome of time to first fall or death was higher in all groups taking higher doses (a statistically significant hazard ratio of 1.54 and a nonsignificant 1.41 for 2,000 and 4,000 IU, respectively). This is why the “best dose” was thus determined to be 1,000 IU per day and the trial moved on from the dose-finding phase to the next phase, which compared 1,000 to 200 IU.
However, this phase didn’t last long, either. Ultimately, the trial was terminated 13 months into the second phase (three years and seven months from the beginning of the study) due to a lack of projected benefit. You might imagine that a trial terminated early due to a lack of effect would have few, if any, statistically significant effects. And you would be right: in the confirmatory stage of this trial, the only difference was the rate of adverse events, which occurred at a higher rate among participants receiving 1,000 IU per day than among those receiving 200 IU per day. Some more details are laid out in Figure 3.
Figure 3: Main outcomes from this study
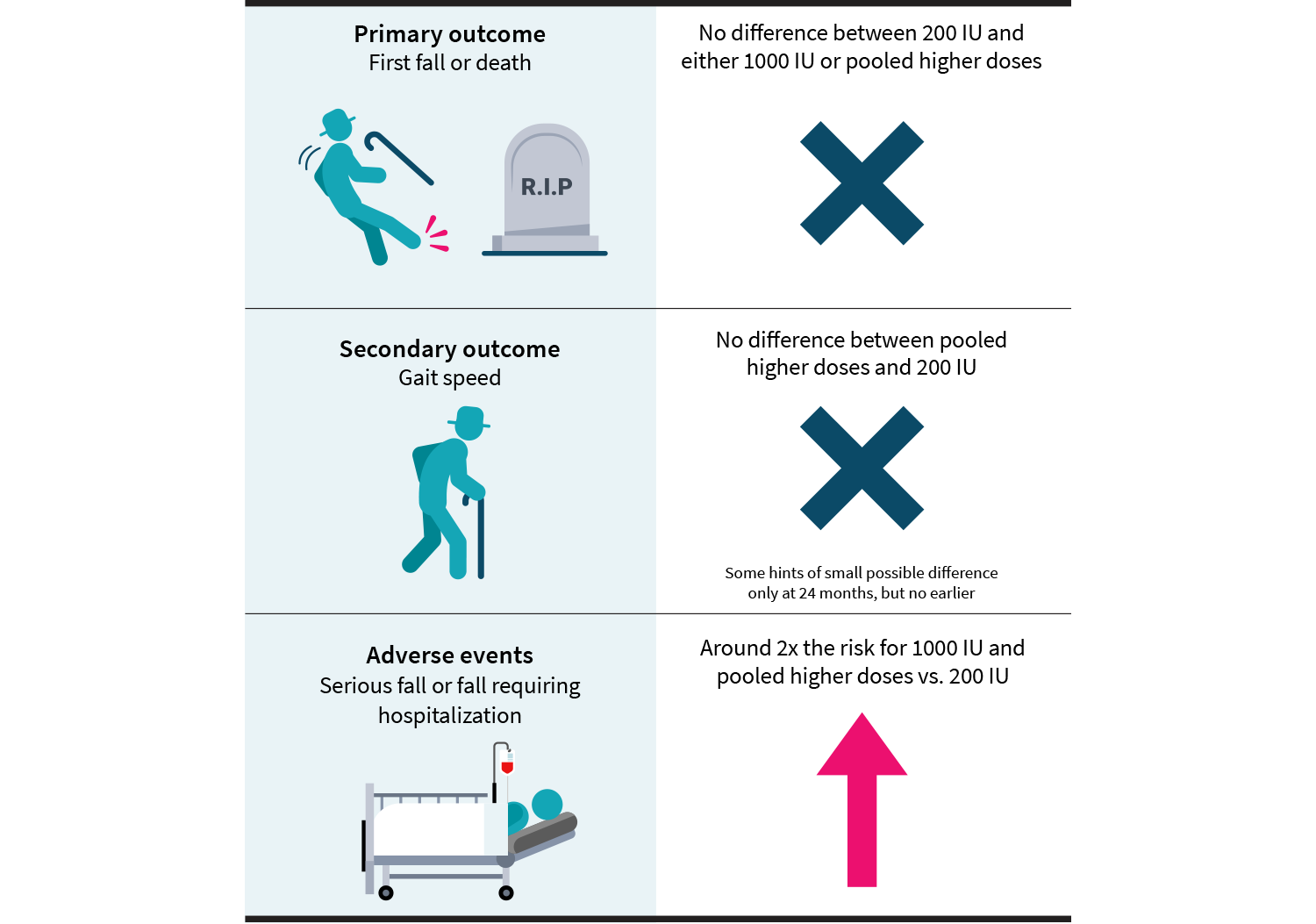
The first half of this adaptive trial suggested that vitamin D doses of 2,000 and 4,000 IU per day are associated with increased risk of falls requiring hospitalization and deaths. At this point, the 2,000 and 4,000 IU per day arms were dropped, and the second phase of the trial continued with just the 200 and 1,000 IU per day arms. The second half of the trial was then halted early due to a lack of effect on falls and an increase in risk for adverse events (hospitalizations from falls, first fall) in the 1,000 IU per day arm.
This trial gives a somewhat clear answer to one question: Does supplementation beyond a low maintenance dose do anything for falls, harmful or beneficial? The answer: Probably not in the context of a moderate dose of 1,000 IU. Though the study provides an answer to one question, it also raises another: does high-dose vitamin D supplementation increase the risk of falls?
Similar results have been hinted at in previous studies. For example, one randomized controlled trial on the effect of high dose vitamin D supplementation and risk of acute respiratory infections showed that high dose vitamin D (100,000 IU per month) may increase the risk of falls[24]. Another trial assessing the effect of high dose vitamin D (60,000 IU per month) on preventing functional decline, showed a similar increase in fall risk[25]. One other dose-response trial reported a U-shaped relationship between vitamin D status and risk of falls. Together, these results suggest a relationship between decreased fall risk with 1,600, 2,400, and 3,200 IU per day, and no decrease in falls with low (400 and 800 IU) vitamin D doses and with high doses (4,000 and 4,800 IU) compared to placebo[26]. One other study looking at the relationship between high dose (4,000 and 10,000 IU per day) vitamin D and bone mineral density suggested that these high doses may in fact decrease bone mineral density[27]. Lastly, during this trial, the 2018 U.S. Preventive Services Task Force reversed its 2012 recommendation, updating their guidelines to say only older adults with osteoporosis or documented vitamin D deficiency should supplement with vitamin D for the prevention of falls[10].
However, not all studies have found a risk of harm. One relevant meta-analysis suggests that there is moderately-certain evidence that vitamin D does not measurably reduce the risk of falls in older adults[28]. Also, a landmark vitamin D study, the VITamin D and OmegA-3 TriaL (VITAL)[29], used higher doses of vitamin D (2000 IU per day) over the course of five years and found no decrease in risk of falls for community dwelling older adults without vitamin D insufficiency and deficiency. Most importantly, though, it did not find an increase in adverse events, either. Additionally, five other related studies report no difference in adverse events between placebo and vitamin D supplementation[15][13][14][30][31]. What could explain these differences?
Most high quality, recent studies take baseline D level into account, so now the most important and often unmeasured variable is a participant’s individual response to vitamin D supplementation[32]. In the present trial, 37.2% of the study participants were already taking a vitamin D supplement (less than 1,000 IU, median 700 IU daily), yet their average serum vitamin D status was still low. One explanation may be that the average BMI of the study population was 30.5, just above the cutoff for obesity. People with obesity are known to have lower vitamin D levels than lean individuals due to a volumetric dilution effect, and it takes a higher dose of vitamin D to raise serum levels to that seen in normal-BMI individuals[33]. Thus, BMI and body fat differences in the study population could account for some of the differences between studies.
There are other hypotheses for why some individuals can take vitamin D on a daily basis and yet still have deficient or insufficient vitamin D levels. One suggests that chronic conditions may have a vitamin D depleting effect[34]. Another theory has to do with genetic polymorphisms of proteins involved in vitamin D metabolism, which may be associated with variability of vitamin D levels after supplementation[35][36] and the incidence of bone diseases[37][38].
Then there’s the issue of the large interindividual variability in response to vitamin D supplements. The same dose of vitamin D in a supplement can produce markedly different increases in serum vitamin D levels[39][40][36] depending on the person (read more in issue #32 of Study Deep Dives here).
The interaction between vitamin D and physical activity is another area not investigated in this study that could help to determine who really benefits from a vitamin D supplement. Previous research has shown that vitamin D receptor (VDR) expression declines with age, but VDR expression increases after an acute bout of exercise, and baseline cardiorespiratory fitness level predicts greater VDR expression. If there was a difference in physical activity among participants in different studies (ie. differences in built environment, severity of self-reported frailty, availability of physical activity programs, differences in living situation, etc.), then this could explain some of the variability across studies.
Finally, the lack of comprehensive data collection prevents the authors from making any educated guesses on why higher doses of daily vitamin D are associated with an increased risk of falls. Specifically, subgroup analyses could have provided a hint as to whether participants with low blood pressure, or participants with lower lean body mass or higher fat mass, or participants with greater functional impairment were more likely to have a negative effect from high-dose vitamin D supplementation. More importantly, this information would help assess whether these potentially confounding attributes were uniformly distributed across the intervention groups.
While these are all possibilities, in the end they’re just guesses. More evidence exploring interindividual differences in vitamin D metabolism could help illuminate the mechanistic underpinnings of the ill effects seen in this study.
This trial suggests that higher doses of vitamin D are not helpful for preventing falls and may in fact increase the chance falls in older at-risk people. This is not the first trial to suggest that vitamin D doses of over 1,000 IU could increase fall risk in older people. However, not all the evidence points toward this risk. More research, focusing especially on individual differences in reactions to vitamin D supplementation, may shed more light on the matter.
Q. Is megadose supplementation a safe practice?
Many physicians provide vitamin D supplements to correct severe vitamin D deficiency through bolus injections of 60,000 once a month or up to 300,000 IU doses every three months until blood levels indicate adequacy. If the “high dose” of 2,000 IU per day (comparable to 60,000 IU per month) incurs an increased risk for falls in this study, it may make sense that megadose supplementation could transiently increase the risk for adverse outcomes due to vitamin D as well.
The researchers behind a recent meta-analysis attempted to find an answer to this question (reported on in the recent January 2019 Mini), but the results were mostly inconclusive. Individual factors introduce so much variability that the answer depends on the person. Vitamin D megadoses may or may not be associated with increased risk of adverse events, kidney stones, and high blood calcium levels. Each of these adverse events could be more concerning for someone with a condition that affects vitamin D metabolism (diseases of the gastrointestinal system, liver, thyroid, kidneys, etc.).
Regular, seasonal serum vitamin D readings help providers determine the ideal dose regimen for an individual. The Food and Nutrition Board (FNB) at the National Academies of Sciences, Engineering, and Medicine (NASEM) recommend that people should avoid serum 25(OH)D levels above 125–150 nmol/L (50–60 ng/mL), and that even lower serum levels (75–120 nmol/L; 30–48 ng/mL) are associated with increased risk of mortality, some cancers (e.g. pancreas and prostate), cardiovascular events, and falls and fractures among older adults[22].
Acute vitamin D poisoning is exceedingly rare. One case report[41] documents a son and his father who had somehow mixed crystalline vitamin D into their table sugar. The pair had been ingesting 1.7 million IUs per day, but even at that dose it took seven months for adverse reactions to precipitate!
The results of this study indicate that, compared to a maintenance dose (200 IU per day) of vitamin D, supplementing with higher doses (1000–4000 IU per day) may result in a higher chance of falls and increased mortality in high-risk community-living adults over 70 years of age.
Not all evidence suggests harm, and these results do not exclude the possibility that higher doses of vitamin D may still be beneficial for some populations. Nor is the harm of higher doses of vitamin D set in stone, given the inconsistent results across different trials. However, given the lack of benefit seen in doses over 1,000 IU per day, coupled with the hint of possible harm, the risks of higher-dose supplementation should be taken into account when considering whether or not to supplement.